by National Institutes of Health
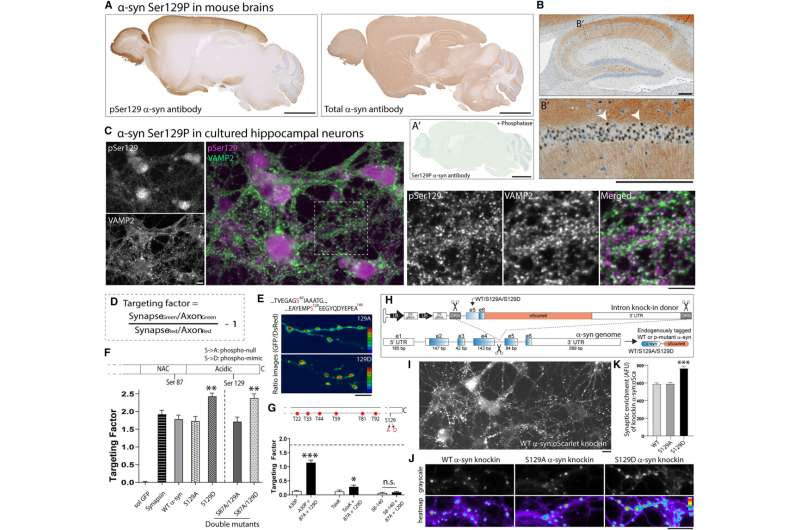
Localization of α-syn Ser129P and effects of phosphorylation on synaptic targeting. Credit: Neuron (2023). DOI: 10.1016/j.neuron.2023.11.020
Researchers have discovered that a protein called phosphorylated α-synuclein, which is associated with several neurodegenerative diseases such as Parkinson’s disease and Lewy body dementia, is also involved in the normal processes of how neurons communicate with each other in a healthy brain.
The work is published in the journal Neuron.
Phosphorylation is a process where a phosphate ion is added to a specific amino acid, or building block, of a protein, in this case the protein α-synuclein. This addition can change the shape of that protein, causing it to change its level of activity.
Most studies of phosphorylated α-synuclein have studied its role in certain neurological disorders such as Parkinson’s disease and Lewy body dementia, where it builds up in protein clumps called Lewy bodies. These clumps are thought to be toxic to neurons, and one of the prevailing hypotheses is that the phosphorylation of the protein α-synuclein triggers these diseases.
“In most studies to date, the mere presence of α-synuclein phosphorylation is assumed to be a marker for pathology for certain disorders, like Parkinson’s and Lewy body dementias,” said Beth-Anne Sieber, Ph.D., program director, NINDS. “Recently, there has been considerable interest in developing drugs that prevent α-synuclein phosphorylation as a way of treating these disorders. These findings challenge the current hypotheses about how these disorders may originate in the brain and may give insight into how we might better treat them.”
Previous work by the lab of Subhojit Roy, M.D., Ph.D., professor at the University of California, San Diego, and senior author of the study, suggested that in a healthy brain, the α-synuclein protein tones down excessive neuronal firing to regulate neuronal communication. While exploring this theory, Roy’s group unexpectedly found that phosphorylation was necessary for the normal function of α-synuclein.
Using a molecular modeling strategy to look at the structure of α-synuclein, Roy and his colleagues led by postdoctoral researcher Leonardo Parra-Rivas, Ph.D. discovered that when α-synuclein is phosphorylated, its structure changes in a way that promotes interactions with other proteins in healthy brains.
Furthermore, they observed an association between increasing neural activity electrically or chemically and an increase in the amount of phosphorylated α-synuclein in both cultured cells and in mouse brain tissue. This finding suggests there might be a relationship between synaptic activity and α-synuclein phosphorylation.
Additionally, experiments show phosphorylation is necessary for α-synuclein to play its role in assembling a network of proteins that bind up synaptic vesicles—pockets that release chemicals enabling neurons to communicate with one another and other cells—and to slow neuronal activity. Therefore, phosphorylated α-synuclein acts almost as a brake or clutch mechanism to keep activity in certain neuronal circuits in check, suggesting that it might have a role in healthy brains, which had not previously been investigated.
“In hindsight, we hadn’t been looking at synuclein phosphorylation the right way,” said Roy. “Take, for instance, the circuits in the olfactory bulb, which according to our data has high levels of phosphorylated α-synuclein. The nose never stops smelling, so it needs to be active all the time. One hypothesis is that synuclein phosphorylation may have evolved as a safety mechanism to protect neuronal circuits that need to be hyperactive.”
The constant presence of α-synuclein phosphorylation in certain brain regions might reflect a need for this biochemical state in those areas. Additional studies are needed to understand how relatively low-frequency events in a healthy brain, when accumulated over a lifetime, can trigger the pathological accumulation of α-synuclein into Lewy bodies, leading to Parkinson’s disease and Lewy body dementias.
Furthermore, therapies designed to block the phosphorylation of α-synuclein itself may need to consider the unintended adverse consequences of blocking a process that may help keep neurons functional during peak activity periods.
More information: Leonardo A. Parra-Rivas et al, Serine-129 phosphorylation of α-synuclein is an activity-dependent trigger for physiologic protein-protein interactions and synaptic function, Neuron (2023). DOI: 10.1016/j.neuron.2023.11.020
Journal information: Neuron

Leave a Reply