by The Mount Sinai Hospital
Graphical abstract. Credit: Cell Reports (2022). DOI: 10.1016/j.celrep.2022.110877
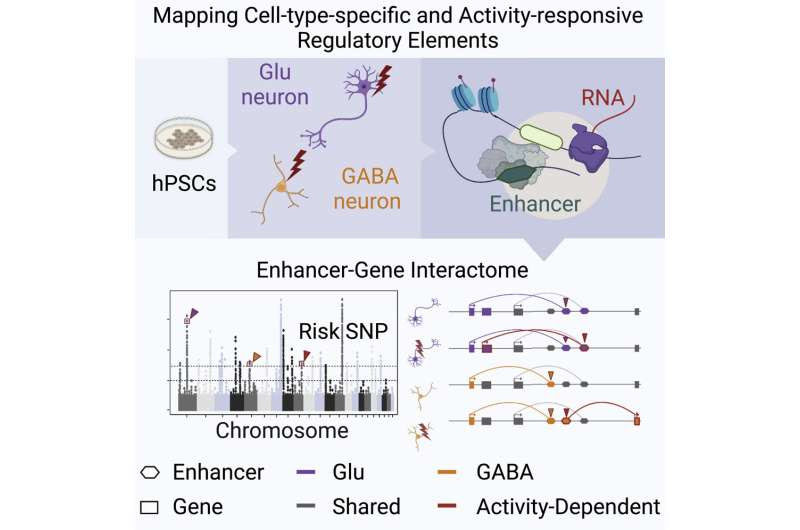
Researchers from the Icahn School of Medicine at Mount Sinai have applied a novel stem cell model to map disease risk variants in human neurons, which could help provide insights into the biological mechanisms that underlie neuropsychiatric disorders such as autism and schizophrenia.
The team’s in vitro cellular model, described in the May 31 issue of Cell Reports, is designed to enable future researchers to elucidate the disease mechanisms involving genome-wide association studies (GWAS) that characterize different risk alleles (common genetic variants conferring risk) for psychiatric disorders. This research could potentially lead to improved diagnostics for the detection of psychiatric disorders years before symptoms appear in patients.
The study focuses on mapping cis-regulatory elements in human neurons that can be linked to psychiatric disease heritability. Cis-regulatory elements, such as promoters and enhancers, are non-coding DNA sequences regulating gene expression and are thus vital components of the genetic regulatory network. Previous genetic studies have revealed a significant enrichment of common variants in the cis-regulatory elements, including those associated with autism spectrum disorder, schizophrenia, and bipolar disorder.
“While common risk variants can shed light on the underlying molecular mechanism, identifying causal variants remains challenging for scientists,” says Nan Yang, Ph.D., Assistant Professor of Neuroscience at the Icahn School of Medicine of Mount Sinai, and senior author of the study. “That’s because cis-regulatory elements, particularly the enhancers, vary across cell types and activity states. Typically, researchers can only use postmortem brain samples where the neurons are no longer active. As a result, they are likely to miss enhancers that only respond to stimulation. Our approach is to map cis-regulatory elements in human neurons derived from pluripotent stem cells. That allows us to replicate neurons in the human brain that can be affected by different types of neuropsychiatric disease, and conduct mechanistic studies of human genetic variants that are inaccessible from other types of human samples.”
In recent years, GWAS have identified hundreds of gene regions associated with psychiatric disease, though understanding disease pathophysiology has been elusive. The functional genomics approach Dr. Yang and her team developed uses stem cell models that can help resolve the impact of patient-specific variants across cell types, genetic background, and environmental conditions. This unique approach effectively lays a foundation to translate risk variants to genes, genes to pathways, and pathways to circuits that reveal the synergistic relationship between disease risk factors within and between the cell types in the brain.
“Our research attempts to decode and transfer highly complex genetic insights into medically actionable information,” says Dr. Yang, who is a member of the Black Family Stem Cell Institute, The Friedman Brain Institute, and The Ronald M. Loeb Center for Alzheimer’s Disease within the Mount Sinai Health System. “That means improving our diagnostic capabilities, predicting clinical trajectories, and identifying pre-symptomatic points of therapeutic intervention for psychiatric disorders.”
By characterizing cell-type specific and activity-regulated gene expression patterns in human cell-derived neurons, Dr. Yang believes her team’s study can greatly benefit the research community. “Our data can guide choosing relevant cell types of experimental conditions to further elucidate molecular mechanisms of disease across the genome,” she points out. “And that could lead to the development of biomarkers that might detect neuropsychiatric disorders years before they manifest themselves in patients, while there is still time to delay or possibly prevent them.”

Leave a Reply