by Salk Institute
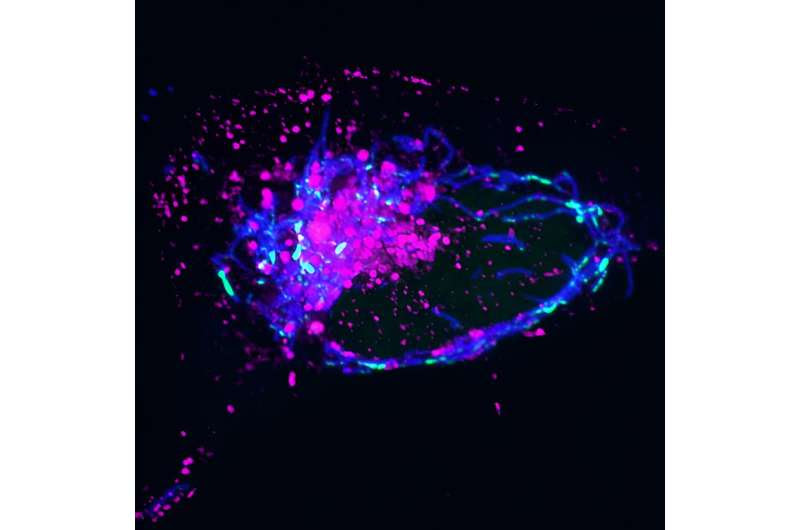
Endosomes (magenta) collect around mitochondria (blue) after infection with virus HSV-1, which attacks mtDNA (green) and causes its release. Credit: Salk Institute
Cells in the human body contain power-generating mitochondria, each with their own mtDNA—a unique set of genetic instructions entirely separate from the cell’s nuclear DNA that mitochondria use to create life-giving energy. When mtDNA remains where it belongs (inside of mitochondria), it sustains both mitochondrial and cellular health—but when it goes where it doesn’t belong, it can initiate an immune response that promotes inflammation.
Now, Salk scientists and collaborators at UC San Diego have discovered a novel mechanism used to remove improperly functioning mtDNA from inside to outside the mitochondria. When this happens, the mtDNA gets flagged as foreign DNA and activates a cellular pathway normally used to promote inflammation to rid the cell of pathogens, like viruses.
The findings, published in Nature Cell Biology, offer many new targets for therapeutics to disrupt the inflammatory pathway and therefore mitigate inflammation during aging and diseases, like lupus or rheumatoid arthritis.
“We knew that mtDNA was escaping mitochondria, but how was still unclear,” says senior and co-corresponding author Professor Gerald Shadel, director of the San Diego-Nathan Shock Center of Excellence in the Basic Biology of Aging and holder of the Audrey Geisel Chair in Biomedical Science at Salk.
“Using imaging and cell biology approaches, we’re able to trace the steps of the pathway for moving mtDNA out of the mitochondria, which we can now try to target with therapeutic interventions to hopefully prevent the resulting inflammation.”
One of the ways our cells respond to damage and infection is with what’s known as the innate immune system. While the innate immune response is the first line of defense against viruses, it can also respond to molecules the body makes that simply resemble pathogens—including misplaced mtDNA. This response can lead to chronic inflammation and contribute to human diseases and aging.
Scientists have been working to uncover how mtDNA leaves mitochondria and triggers the innate immune response, but the previously characterized pathways did not apply to the unique mtDNA stress conditions the Salk team was investigating. So, they turned to sophisticated imaging techniques to gather clues about where and when things were going awry in those mitochondria.
“We had a huge breakthrough when we saw that mtDNA was inside of a mysterious membrane structure once it left mitochondria—after assembling all of the puzzle pieces, we realized that structure was an endosome,” says first author Laura Newman, a former postdoctoral researcher in Shadel’s lab and current assistant professor at the University of Virginia.
“That discovery eventually led us to the realization that the mtDNA was being disposed of and, in the process, some of it was leaking out.”
The team discovered a process beginning with a malfunction in mtDNA replication that caused mtDNA-containing protein masses called nucleoids to pile up inside of mitochondria. Noticing this malfunction, the cell then begins to remove the replication-halting nucleoids by transporting them to endosomes, a collection of organelles that sort and send cellular material for permanent removal.
The endosome gets overloaded with these nucleoids, springs a leak, and mtDNA is suddenly loose in the cell. The cell flags that mtDNA as foreign DNA—the same way it flags a virus’s DNA—and initiates the DNA-sensing cGAS-STING pathway to cause inflammation.
More information: Mitochondrial DNA replication stress enacts an endosomal pathway of nucleoid disposal prone to innate immune system activation, Nature Cell Biology (2024). DOI: 10.1038/s41556-023-01343-1. www.nature.com/articles/s41556-023-01343-1
Journal information: Nature Cell Biology
Provided by Salk Institute

Leave a Reply