by Perelman School of Medicine at the University of Pennsylvania
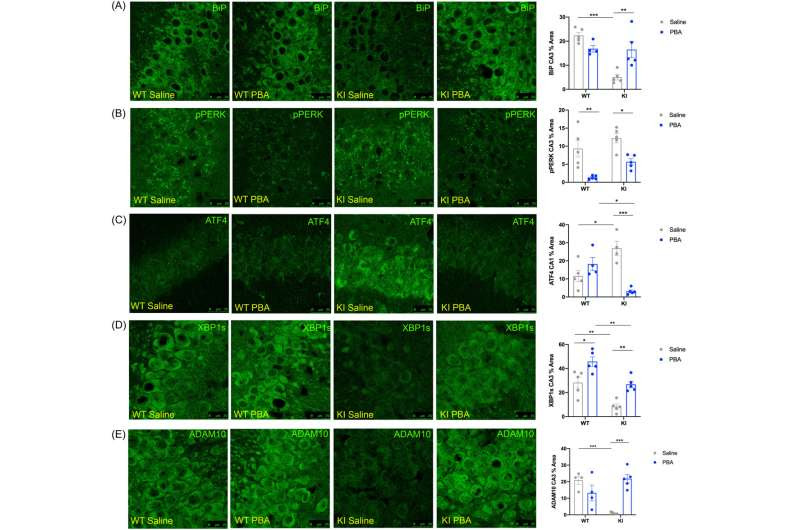
PBA treatment reduces PERK activity and increases XBP1s in the hippocampus of APPNL-G-F knock-in (KI) mice. Confocal images of the hippocampus across groups. (A) BiP in the CA3. (B) p-PERK in the CA3. (C) ATF4 in the CA1. (D) XBP1s in the CA3. (E) ADAM10 in the CA3. Data quantified and presented as mean ± SE percent area of BiP, p-PERK, ATF4, XBP1s, and ADAM10 within hippocampal sections (n = 4–5 animals per group; two-way ANOVA with Tukey post hoc correction for multiple comparisons, *p < 0.05, **p < 0.01, ***p < 0.001). Credit: Aging Biology (2023). https://agingcelljournal.org/Archive/Volume3/20230017/
A “chaperone” molecule that slows the formation of certain proteins reversed disease signs, including memory impairment, in a mouse model of Alzheimer’s disease, according to a study from researchers at the Perelman School of Medicine at the University of Pennsylvania.
In the study, published in Aging Biology, researchers examined the effects of a compound called 4-phenylbutyrate (PBA), a fatty-acid molecule known to work as a “chemical chaperone” that inhibits protein accumulation. In mice that model Alzheimer’s disease, injections of PBA helped to restore signs of normal proteostasis (the protein regulation process) in the animals’ brains while also dramatically improving their performance on a standard memory test, even when administered late in the disease course.
“By generally improving neuronal and cellular health, we can mitigate or delay disease progression,” said study senior author Nirinjini Naidoo, Ph.D., a research associate professor of Sleep Medicine. “In addition, reducing proteotoxicity—irreparable damage to the cell that is caused by an accumulation of impaired and misfolded proteins—can help improve some previously lost brain functions.”
Alzheimer’s disease impacts more than 6 million Americans, and as many as 13.8 million Americans could be diagnosed by 2060, barring medical breakthroughs to slow or cure the disease. Like other neurodegenerative disorders, Alzheimer’s disease features the accumulation of protein aggregates in the brain, and includes the dysfunction of proteostasis, itself.
Previously, researchers found that PBA treatment improved sleep quality and cognitive test performance—and helped normalize proteostasis—in mice that model ordinary human brain aging. For the new study, they investigated PBA’s effects in mice that model Alzheimer’s disease. These mice, known as APPNL-G-F mice, accumulate abnormal protein aggregates in their brains, lose many of the synapses that connect their brain cells, and develop severe memory impairment—much like people with Alzheimer’s.
First, the team showed that these mice do indeed have signs of dysfunctional proteostasis mechanisms—including a chronically activated process called the unfolded protein response—and relatively low levels of a natural aggregate-preventing “chaperone” protein called binding immunoglobulin protein (BiP) or Hspa5.
Next, graduate student Jennifer Hafycz treated the mice, starting early in life, with PBA, finding that the treatment helped restore signs of normal proteostasis in key memory-related brain regions in the mice. The treatment also restored the mice’s ability—which was otherwise abolished—to discriminate between moved and unmoved objects on a standard memory test called the Spatial Object Recognition test.
The team discovered that they could achieve similar effects, including the reversal of memory deficits, even when they treated the mice starting in middle age.
Both early life and middle-age treatment showed signs of inhibiting the process that forms the most prominent protein aggregates in Alzheimer’s, known as amyloid beta plaques. For the later treatment, not only the underlying process but also the amyloid plaque numbers themselves were reduced.
As a potential Alzheimer’s treatment, PBA has the advantage that it can cross easily from the bloodstream into the brain and is already approved by the Food and Drug Administration for treating an unrelated metabolic disorder.
More information: Jennifer M. Hafycz et al, Early and Late Chaperone Intervention Therapy Boosts XBP1s and ADAM10, Restores Proteostasis, and Rescues Learning in Alzheimer’s Disease Mice, Aging Biology (2023). agingcelljournal.org/Archive/Volume3/20230017/
Provided by Perelman School of Medicine at the University of Pennsylvania

Leave a Reply