Peer-Reviewed Publication
TAMPERE UNIVERSITY
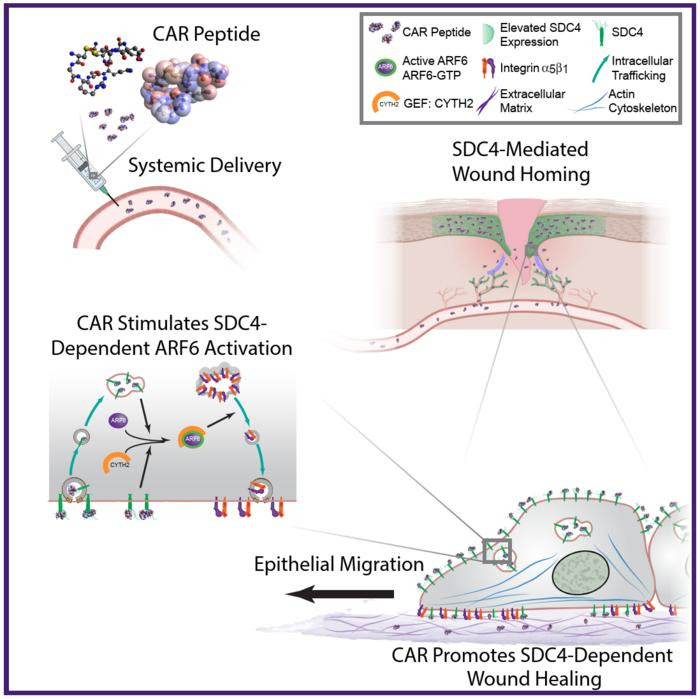
SCHEMATIC DIAGRAM HIGHLIGHTING PROPOSED MECHANISM OF ACTION OF CAR PEPTIDE. SYSTEMICALLY ADMINISTERED CAR PEPTIDE ASSOCIATES WITH THE HSPG SDC4, WHICH IS RESTRICTED TO EPIDERMIS AND BLOOD VESSELS IN MOUSE SKIN WOUNDS. CAR INDUCES SDC4-DEPENDENT ACTIVATION OF THE SMALL GTPASE ARF6, VIA THE GUANINE NUCLEOTIDE EXCHANGE FACTOR CYTH2, TO PROMOTE SDC4-, ARF6- AND CYTH2-MEDIATED KERATINOCYTE MIGRATION AND ENDOGENOUS RE-EPITHELIALISATION AND WOUND REPAIR MECHANISMS.
CREDIT: TAMPERE UNIVERSITY
One of the key goals of medical science is to speed up the healing of tissue injuries in a way that would not enable the forming of less functional scar tissue in the affected areas. Professor Tero Järvinen at Tampere University in Finland is leading research that is seeking solutions to achieve this goal.
In an earlier study, Järvinen discovered a so-called homing peptide, a small piece of protein that specifically seeks tissue damage via the bloodstream. The homing peptide was named the CAR peptide after its amino acid composition. It was screened out of more than a billion peptides using the screening technology of in vivo phage display. The peptide was used to transport a scar reducing drug to the wounds via systemic administration.
Recently, the research group showed that the CAR transport peptide is itself a therapeutic drug. CAR activates a natural healing pathway crucial for tissue regeneration and accelerates wound healing without the need to attach to any kind of drug.
The study, reported in a recent issue of Nature Communications, confirmed that the CAR peptide activates the tissue regeneration mechanism that is critical for wound healing by entering the wound through the bloodstream. In the wound, the peptide binds to syndecan-4, a cell membrane receptor. This binding activates the intracellular cytohesin-2 factor, which in turn activates the Arf6 GTPase that triggers cell migration, i.e., the migration of regenerating cells across the damaged area in the wound.
By this molecular mechanism, the skin wounds in mice receiving the CAR peptide healed significantly faster than without the therapy. The ensuing scars were also smaller than without treatment. As this syndecan-4 dependent cell migration-mechanism is crucial for the healing of multiple injuries, such as skeletal muscle ruptures and bone fractures, the CAR peptide has multiple clinically relevant indications not just for traumatology, but also surgery and sports medicine.
“Syndecan-4-dependent tissue regeneration is a key factor in the healing of several other tissue injuries beyond skin wounds, including muscle ruptures and bone fractures. The CAR peptide we found, which activates this natural healing mechanism, opens completely new possibilities for treating traumas and injuries not only in traumatology but also in surgery and sports injuries. Because the drug molecule actively seeks tissue damage through the bloodstream, the treated injury can be in any part of the body and can be targeted by the CAR peptide therapy,” Järvinen explains.
Professor Tero Järvinen led the study in cooperation with Academician of Science Erkki Ruoslahti. The research was carried out at Tampere University, Sanford Burnham Prebys Medical Discovery Institute (La Jolla, CA, USA) and at the University of California Santa Barbara (UCSB, CA, USA). Researchers from the University of Liverpool (UK) and the Wellcome Trust Centre for Cell-Matrix Research at the University of Manchester (UK) also participated.
The research article was published in Nature Communications.
JOURNAL
Nature Communications
DOI
10.1038/s41467-023-43848-1
ARTICLE TITLE
Systemically administered wound-homing peptide accelerates wound healing by modulating syndecan-4 function
ARTICLE PUBLICATION DATE
6-Dec-2023

Leave a Reply